Unit 6 - Bonding
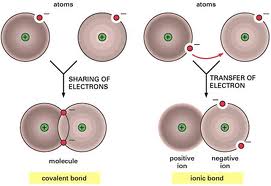
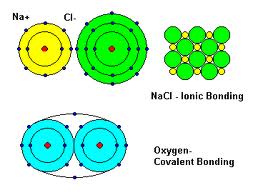
In nature, atoms of almost all the elements on the periodic table are found in combination with other atoms. When atoms of one element combine wiht atoms of a different element, they form a compound. In an ionic compound, one or more electrons are transferred from the aoms of metals to atoms of nonmetals. In compounds of non metals, covalent bonding occurs by atoms sharing one or more valence electrons. There are many more covalent compounds than there are ionic ones!!!
MaterialsBonding Lab
Bonding Lab Data Table Bonding PPT & Notes Types of Bonding WKST Types of Bonding WKST Answers Electron-Dot Formula Notes Electron-Dot Formulas WKST
Electron-Dot Formulas WKST Answers Building Molecular Models Activity Electron Dot (ED), Shapes, and Polarity WKST Electron Dot (ED), Shapes, and Polarity WKST Answers Electronegativity & Polarity PPT & Notes Electronegativity & Polarity Packet Electronegativity & Polarity Packet Answers Unit 6 Chemical Bonding Test Review Unit 6 Chemical Bonding Test Review Answers Chemical Bonding Jeopardy |
Reinforcements |